|
1/17/2024 0 Comments Changing states of matterAs a result, solids have the lowest kinetic energy of all the states of matter. In other words, they stay in their fixed positions. Due to the strong forces holding them together, the particles of a solid are only able to move back and forth in small vibrations. 
In the solid state, particles tightly pack together in a fixed arrangement. Additionally, there are many intermediate states, many of which only exist under extreme conditions in total, there are twenty! Due to their difference in properties being their distinguishing factors, let’s go over some of the states of matter below: Solids There are four main states of matter if you include plasma. The properties of a substance, such as its density, conductivity, and viscosity, also vary depending on its state of matter. Solids have a fixed shape and volume, while liquids have a fixed volume but can take the shape of their container, and gases have neither a fixed shape nor a fixed volume. The behavior of matter also changes depending on its state. But if the water is heated to a high enough temperature, it will become a gas (steam), and if it is cooled to a low enough temperature, it will become a solid (ice). For example, at room temperature and pressure, water is a liquid. The state of a substance depends on its temperature and pressure. There are three main states of matter: solid, liquid, and gas. The states of matter refer to the physical forms that matter can take. Phase and Matter are occasionally used as synonyms however, it is possible to form several phases that are in the same state of matter (such as how solids can have different crystalline structures).For more in-depth information on the concept of matter, check out this article! Matter– anything that has mass and occupies space it constitutes atoms and compounds, which compose physical and chemical properties.A moving electric charge in a magnetic field experiences a force perpendicular to its velocity. Magnetic field – a region around a magnet or an electric current that describes the magnetic influence on moving electric charges, currents, and magnetic materials.Properties of Solids, Liquids, and Gases.The heat energy needed to warm or cool solids, liquids, and gases without changing phase is also accurately known.In this tutorial, you will learn about the four main states of matter (solid, liquid, gas, and plasma), as well as some intermediate states of matter, by reading about their properties, applications, and examples. The energy involved in phase changes is accurately known for many substances. As solid water converts to liquid water, heat is absorbed as the forces between water molecules weaken, allowing the liquid to flow. Because phase diagrams provide so much information, they are known for thousands of substances.Īny change in phase is accompanied by the taking in or release of heat energy because, as change takes place, the attractive forces between molecules are being broken down or being formed. The relationship between temperature and pressure and the three states of matter is easier to see when displayed in a phase diagram. Changes in state can also occur by changing the pressure while holding temperature constant. Going the opposite direction in temperature and at this same pressure, water changes to a gas when the temperature exceeds 100☌. But if its temperature is lowered to –0.01☌, liquid water freezes to solid ice. At room temperature (about 22☌) and at the normal pressure exerted by the atmosphere, water exists as a liquid, which can flow from one container to another. Whether a substance is a solid, a liquid, or a gas depends on its temperature and the pressure placed on it. These are the three common states of matter. Nearly every substance can exist as a solid, a liquid, or a gas. Quiz: Introduction to Oxidation-Reduction Reactions.Introduction to Oxidation-Reduction Reactions.Quiz: Heat Capacities and Transformations.Quiz: Introduction to Organic Compounds. 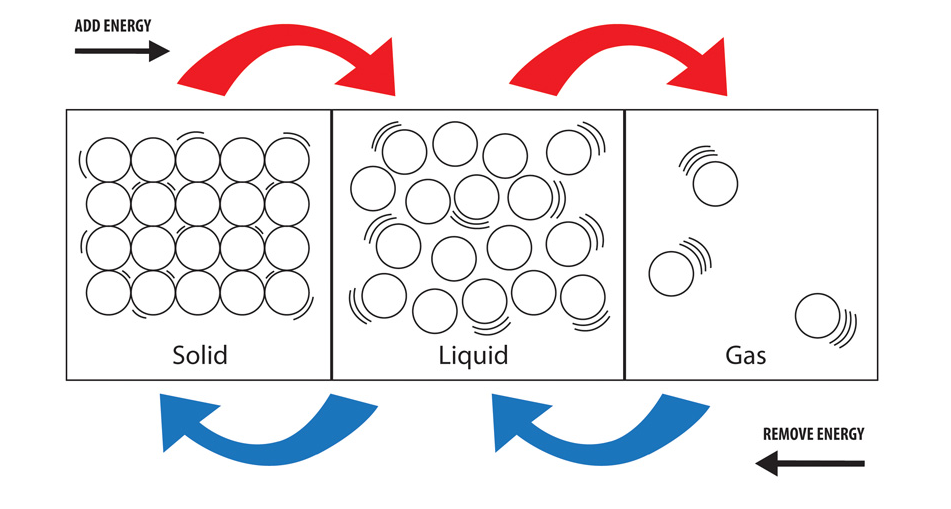
Quiz: Compounds with Additional Elements.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
